Family Trees: Roots & Resilience
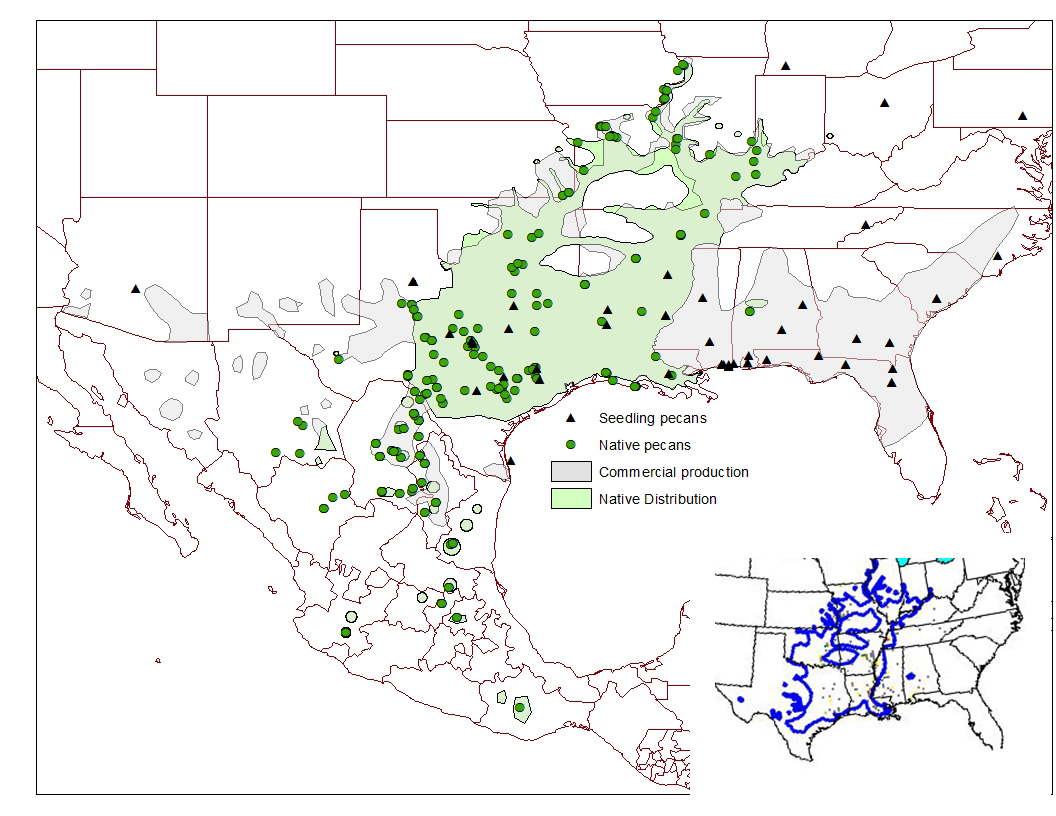
Figure 1: Areas of native and commercial pecan distribution in the U.S. and Mexico, with points of origin for selected individual native and seedling accessions in the NCGR-Carya. Insert is Little's (1971) map.
In the March edition of Pecan South, I told stories of the origins of some important cultivars for the western region, ‘Western’ and ‘Riverside’, and tied those stories to currently developing techniques of genetic analysis. Now, I want to turn to the story of Florida pecans, whose stories are embedded in the nursery industry as rootstocks as well as scion cultivars and parents in breeding and development.
As before, these are the stories of our pecan growing families with direct links to our current industry, and these stories cross national borders that are still in contention. Furthermore, these stories highlight the role of plant introductions in developing regionally adapted cultivars. They also have implications for our use of diversity in the USDA ARS National Collection of Genetic Resources for Pecans and Hickories (NCGR-Carya) for cooperatively breeding and selecting better cultivars and rootstocks in the future. Accuracy requires documentation, so references are provided. Researchers are an important part of our pecan family, and documentation increases communication between our research teams and the industry we serve.
Native pecan distribution
Previous plant collectors’ records are valuable when searching for a plant in a new area. Valuable records are found in herbarium vouchers (pressed plant specimens) that botanists have made when documenting their collections and findings. Records of those collections are compiled across many herbaria to create distribution maps, such as were developed for the original native pecan forest by E. L. Little (1971) that were based on herbarium records and historical accounts. The original native pecan forest extended down the Mississippi Valley, sometimes on both sides of the river, to Louisiana. Isolated populations that were separated from the main group were reported from Alabama (Mohr, 1893; Harper, 1928) and Ohio (Braun, 1961) but the main forest did not extend into the eastern states of Florida, Georgia, the Carolinas, or Virginia.
Native distribution is important to consider when evaluating eastern pecan cultivars; those cultivars outside of the native distribution indicate plant introductions, raising questions of where the seed originated. I have trusted Little’s pecan distribution map in eastern and northern regions but not in West Texas, where native trees are much more widely distributed than his map indicated (Fig 1). It has also been necessary to add estimates of pecan’s native distribution in Mexico since Little’s maps were only for the United States. Pecan distribution in Mexico is based on both plant collection reports like those by Manning (1949) and on records provided by numerous colleagues like Ruben Castro and Humberto Nuñez in the course of working in the area.
Florida Seedstocks
My first interest in pecan cultivars from Florida began in 1982 when I went to Louisiana to work at the Louisiana State University Pecan Research Extension Station. I began a project on rootstock impact on tree performance. Some of the first research done on pecan rootstocks was by Sitton and Dodge (1938) while working at the LSU Pecan Station at Robson, Louisiana when it had been a USDA worksite. They found increased tree size and nut production when ‘Schley’ was grafted onto ‘Moore’ seedling rootstocks, as compared to being grafted on ‘Waukeenah’ seedlings. They also reported decreased size, yield, and iron chlorosis when water hickory (Carya aquatica) was used as a rootstock. I knew that pecan scions could be successfully grafted on all species of Carya; I intended to look at site adaptation of species as they relate to rootstock use, hoping for tree size control. North Louisiana includes most North American species of Carya and was a good place to begin my study of hickories.
I contacted prominent nursery producers in pecan growing states to learn what they used as seed and why. Gast and Overcash (1980) had already provided details of regional seedstock usage in their exhaustive survey—they contacted 73 nurseries and 37 responded! Their survey reported dramatic differences in seed selection by nurseries in different regions but discounted edaphic or climatic adaptation as a cause. However, the most commonly used seedstocks in the eastern region (Florida, Georgia, Alabama, and Mississippi) were ‘Curtis’ and ‘Elliott’, both of which originated in Florida. The most commonly used seedstocks in the western region (Texas, New Mexico, and California) were ‘Riverside’ and ‘Burkett’, both Texas natives. The most commonly used seedstock in Louisiana and Oklahoma, according to Gast and Overcash (1980) was ‘Moore’ (also a Florida seedling selection). Most of the nursery producers I talked with made seedstock selections based on local availability of good quality seed, with uniformity and vigor of seedling establishment being key to continued use. Some were adamant that the choice of seed did not matter.
I obtained open-pollinated seed of seven seedstocks (‘Apache’, ‘Burkett’, ‘Curtis’, ‘Elliott’, ‘Moore’, ‘Riverside’, and ‘Sioux’) and established a nursery in a randomized complete-block design where seedlings were grown and grafted to ‘Candy’, ‘Cape Fear’, or ‘Stuart’. Significant differences were later reported: ‘Curtis’ and ‘Elliott’ rootstocks forced earlier bud growth in scions which had initiated growth and experienced increased damage to a late spring freeze (Grauke and Pratt, 1992). I speculated that the geographic origin of the cultivars used as seed was important to their performance and related to the regions where they performed best. However, ‘Curtis’, ‘Elliott’, ‘Moore’ and ‘Waukeenah’ all originated from Florida. Since pecan was not native to Florida, where did they come from? Blackmon (1927) told the story of the oldest individual pecan tree in Florida (“a seedling on the farm of Mr. Frank Gray of Jacksonville, said to have been planted by the Spaniards in 1825”) and the oldest plantation in the state “made by John Hunt of Bagdad, probably about 1848.”
John Hunt operated a brickyard in Bagdad, Florida as early as 1825 (King, M.L. 1973). Arthur Brown purchased the land from John Hunt in 1883, on which there were 38 large seedling pecan trees. Blackmon (1927) interviewed Arthur Brown’s son J.L. Brown in 1924 to learn more about the origin of Hunt’s pecan trees:
“the only information obtainable was from the former slave and body servant of Mr. John Hunt. The servant, who claimed to be one hundred years old at the time, told the Browns that ‘Mars John’ went to the war in a ship and took his body servant with him. The ship passed in and up a river, but which one [the former slave] did not know. There were numerous pecan trees growing along the banks, some of them producing large nuts of such fine flavor and quality that the servant was required to gather a quantity. Many of the nuts were brought to Florida by Hunt and planted on his farm at Bagdad.”

Figure 3: USDA Logbook entry of pecan samples from Arthur Brown, Bagdad, Florida, received on Nov. 23, 1893.
Among the 38 seedling pecan trees in the Bagdad planting was one of particular size and quality. Arthur Brown exhibited specimens of the nuts from that tree at the New Orleans Exposition (1884) and Paris Exposition (1889) and won prizes for size and quality. Interest in pecans was stimulated to such an extent that Brown sent seed for planting to Durban, South Africa; Queensland, Australia; and Ghorakhal, India. At the time of his writing, Blackmon was confident that the plantings made in those countries were still in existence, based on correspondence with officials in the Departments of Agriculture in the respective countries. In the same time period, Brown sold ‘Turkey Egg’ seed to J.B. Curtis in 1886, from which the ‘Curtis’ pecan cultivar was selected (Taylor, 1907) as well as the cultivars ‘Randall’, ‘Hume’ and ‘Kennedy’ (Thompson and Young, 1985).
Arthur Brown also sent nut samples from his best trees to the USDA Plant Introduction Station, where they were described and saved. When I photographed the nut vouchers in the McKay Nut Collection, I found samples of ‘Turkey Egg Jr’, ‘Turkey Egg Sr’, ‘Repton’, ‘Ribera’ and ‘Harcourt’, all obtained from Arthur Brown in November 1893. The Log Sheet on page 86 described the evaluation of the staff, indicating that ‘Harcourt’ was “probably the best of the lot.” ‘Turkey Egg Sr.’ was the largest nut, but kernels were “a little tough… not the highest quality.”
Elliott and Curtis pecans
Pictures of the 1893 crop of nuts from Arthur Brown’s orchard are a link back the plant introductions that John Hunt made to Bagdad, Florida when he planted the nuts his servant collected in 1848. Most pecan growers who are familiar with ‘Elliott’ and ‘Curtis’ will see similarities between Brown’s nut samples and those two modern cultivars. Taylor’s (1907) account of the origin of ‘Curtis’ ties it to the ‘Turkey Egg’ seed sold to J.B. Curtis by Arthur Brown. The original ‘Curtis’ from the 1906 crop of J.B. Curtis in the McKay Nut Collection is typical of the grafted cultivar being grown in orchards today.
The linkage between the John Hunt orchard at Bagdad, and the pecan cultivar we know as “Elliott” is more challenging but is important. ‘Elliott’ is a seedling selection made by Effie McLean Elliot, the wife of Henry Elliot. Although the family spells its name with one “t,” the cultivar became established in the industry spelled with two “t’s” as shown in Thompson and Young (1985). The tree was 2.5 feet in diameter and known for making large crops of high-quality nuts when the family purchased the property where it stood in Milton, Florida (Arnold, 1971). According to her granddaughter Pamela Elliot Reinhart, Effie was responsible for making the selection and working to have the name registered in 1912, the year that Henry died. The town of Milton is located in Township 1N, Range 28W of Santa Rosa County, near the confluence of Pond Creek and the Blackwater River where John Hunt owned land (Vickrey, 1998). The first propagation of the Elliot tree occurred in 1919 with the establishment of Harlan Farms Nursery in Paxton, Florida. The first living inventory of ‘Elliott’ established at the USDA ARS Pecan Breeding Program was grafted in 1952 to BRW 42-28 and the first crosses using its pollen were made the next year.
Louis Romberg, the first USDA pecan breeder, noted that ‘Elliott’ was “a variety from Florida that is gaining in favor because it bears an excellent nut for shelling and the tree has been highly resistant to scab. The foliage of the tree resembles that of Curtis; therefore it appears that it may be a seedling of that variety or otherwise related to it. The tree is one of the first to leaf in spring and one of the first to be receptive for pollination. It lacks precocity and is a moderate bearer. The nut is small to medium in size, roundish, the shell is medium but the parallel grooves are small and there is little of a central partition septum. We consider this variety to be a good parent.”
The diameter of the original Elliot tree in 1912 indicates it grew to maturity prior to ‘Curtis’ and could not be its progeny. If not one of Hunt’s original 38 trees, it probably descended from their seed. ‘Curtis’ was grown from a seed planted in 1886, first fruited in 1893, and was first propagated in 1896 (Taylor, 1907). The original Elliot tree was 2.5 ft diameter when it was discovered in 1912 and produced up to 250 pounds of good quality pecans (Arnold, 1971). If it grew an average of a half inch in diameter per year, it would take 60 years to reach 30 inches. We can then estimate that it was a seed around 1852, which is close to the original Hunt orchard’s establishment.
The picture of the ‘Curtis’ and ‘Elliott’ samples from living inventories of NCGR-Carya are typical of the cultivars. The verified inventories shown here are becoming what botanists refer to as “types:” they can be trusted to represent the cultivar. The genomic profile of verified inventories of ‘Curtis’ and ‘Elliott’ have been published (Marquard et al., 1995; Bentley et al., 2019). By including “populations” of trees from across a wide range in our studies, we improve our methods, which then also show patterns related to groups from certain regions. Some of those patterns will relate to adaptations important for improved performance. Recognizing those patterns leads to genetic “markers” that increase the power and reduce the time needed for breeding and selection.
Plant enzyme analyses indicate that ‘Elliott’ and ‘Curtis’ each have unusual profiles but could have a common parent (Marquard et al., 1995). Microsatellite analysis also shows that they could have the same seed parent (Grauke, unreported data). Romberg noted leaf morphology that connected ‘Elliott’ to ‘Curtis.’ Furthermore, seasonal patterns of growth suggest the two cultivars come from the same “population” (Grauke and Pratt, 1992). Early growth in spring is consistently related to seedlings coming from the southern provenance (Wood et al., 1998; Grauke and Starr, 2014; Bock et al., 2016). ‘Randall’ and ‘Kennedy’, known progeny of ‘Turkey Egg’, and siblings of ‘Curtis’ have recently been added to NCGR-Carya collections and will be profiled using new genomic techniques. Patterns in the siblings should allow us to delineate the profile of their common parent, which was evidently ‘Turkey Egg.’
A pecan tree on its own roots is long-lived and may survive over 200 years, as Romberg carefully documented in his letter to C.D. Raby tracing a tree from 1720 to 1929. Pecan cultivars are “immortal” in that graftwood moved from an ancient tree to a young tree continues to survive. However, not every tree that is called by a cultivar name is a reliable example. NCGR-Carya maintains verified inventories, and the archived genomic profiles for a given cultivar are useful to the industry as a tool to verify identity. The development of grafting was an important point for our developing industry and allowed a single cultivar to be propagated for the production of a uniform crop of large nuts. However, genetic diversity is the foundation of breeding and is the key to resilience in any population.
The USDA ARS Pecan Breeding Program was initially designated as a National Clonal Germplasm Repository based on an existing collection of grafted cultivars developed for use as parents by the USDA ARS Pecan Breeding Program. Breeders recognized the value of maintaining a collection of named cultivars from culturally distinctive growing regions to represent selections made over the 170-year history of grafting pecans.
Pecan researchers in the first Crop Germplasm Committee also saw the need to develop provenance collections of self-rooted seedlings to represent genetic diversity in the context of geographic origin. We worked with colleagues in Mexico to make collections across the U.S. and Mexico to establish orchards in Texas and Georgia (Grauke et al., 1989; Wood et al., 1998;). Those collections and representative collections of Carya species provide the broadest base for studying genetic adaptations to soils and climates, and for breeding pecans.
The broad genetic diversity available in the collection is being used by multiple international teams for developing improved methods of molecular characterization based on genome sequencing (Bentley et al., 2019). Over 400 trees in our collection that originated in locations shown on the earlier map will be sampled in May 2019 for genomic resequencing, providing the complete genomic profile of each plant and the ultimate evidence of its identity within the community. Those genomic profiles will answer questions of genetic relatedness between ‘Elliott’ and ‘Curtis’—possibly identifying the location of the river in South Texas or Mexico where nuts were collected for the first Florida plantations. The comprehensive genomic profiles should illuminate critical adaptations for regional selection: for disease resistance in the East, drought and salt tolerance in the West, and cold hardiness in the North.
Will breeders attempt to develop scion cultivars that can go anywhere or target adaptation to local constraints? Will rootstocks continue to be ignored, will they become the target of independent regional selections that are complementary to scion development, or will trees be assembled to produce seed that can be grown on their own roots? The pecan industry is at a dynamic point in its development. The future will build on the genetically diverse and resilient foundations of the past and make great progress in the future as we develop a historical and genetic understanding of this valuable tree.









